Roadmap Overview
Finding a cure
CureGRIN’s three-year plan focuses on 10 essential research questions that need to be answered in order to find cures and treatments for people living with GRIN Disorder.
Based on input from over 200 families, physicians and researchers, the plan outlines how we’ll work collaboratively with families, researchers, doctors, and industry to explore gene therapy and drugs as potential treatments.
We’ve titled the plan “Treat the Symptoms; Cure the Disease” to reflect input from GRIN families that we should balance efforts to find a cure, with research into symptom-specific treatments that can provide relief over the shorter term.
10 Essential Questions
We’ve identified 10 essential questions that will define what research projects we invest in over the next three years. We hope the questions will also influence how researchers, biotechnology companies, and other GRIN advocacy organizations prioritize their own work.
The 10 Essential Questions are:
Essential Question 1: What are the right outcome measures?
CureGRIN’s Research Roadmap Explained
Each Thursday throughout the summer, CureGRIN will be sharing a blog post that summarizes one of the 10 “Essential Questions” that need to be answered in order to find cures and treatments for GRIN Disorder.
Today, we focus on Question 1: What Are the Right Out Come Measures?
An “outcome measure” is simply a tool that reflects the impact of a treatment or intervention on the health of GRI patients. Outcome measures provide data on the measurable changes in the health or quality of life of patients. We want to make sure that researchers are measuring the things that are most important to us. By agreeing a set of outcome measures, research from different studies can all be compared together.
Some examples of outcome measures include things like seizure frequency or severity and various assessment scores. For an outcome measure of seizure frequency or severity, researchers would measure the “baseline” frequency or severity of seizures (before any treatment is given). Then, researchers would assess the frequency or severity of seizures over a period of time after treatment is given to see if there is a reduction in the frequency or severity of seizures as a result of the treatment. Assessment scores could include things like the Vineland test, which measures behavior for intellectual and developmental disabilities, the Wechsler Adult Intelligence Scale (WAIS), which measures cognitive ability in older adolescents and adults, and the Social Communication Questionnaire, which measures autism spectrum disorder symptomatology. These assessments would be administered pre- and post-treatment to see if the scores improve as a result of treatment.
Some outcome measures being assessed in the Barcelona GRIN Team’s ongoing L-serine clinical trial are the Vineland Adaptive Behavior Scales, Bayley Scales of Infant and Toddler Development, Wechsler Intelligence Scale, Gross Motor Function Measure-88, Social Communication Questionnaire, Sleep Disturbance Scale for Children, and microbiota composition, among others.
Through our participation in CombinedBrain, CureGRIN and GRIN2B Foundation will be teaming up with researchers at Duke University to explore an outcome assessment tool called ORCA. We will work with families, physicians and researchers to determine what the best outcome measures to see benefit from treatments are and how should these be measured for future pre-clinical studies and clinical trials.
Essential Question 2: Can we find biomarkers?
Each Thursday throughout the summer, CureGRIN will be sharing a blog post that summarizes one of the 10 “Essential Questions” that need to be answered in order to find cures and treatments for GRIN Disorder.
Today, we focus on Essential Question 2: Can we find biomarkers?
What if we could find a biomarker that’s only present in the blood of people with GRIN? This might mean that the disease could be diagnosed through a simple blood test. Additionally, measuring that biomarker after a drug or gene therapy could also become an early sign that the treatment is working.
A “biomarker” is something biological, chemical, or physical in nature that can be measured in the body. Biomarkers can be important indicators of normal biological processes and disease state processes. They can also indicate certain responses to medicines or other treatments. Some common examples of biomarkers to think about are things like blood sugar, blood pressure, or the presence and size of a tumor. Blood sugar or blood glucose is a molecular biomarker that can be tested to help physicians understand someone’s risk for diseases like diabetes. Blood pressure is a physiological biomarker that can help physicians monitor the pressure in someone’s arteries when their heart beats and when it rests between beats to assess their risk for high blood pressure (hypertension) and other medical conditions. If someone has a tumor, different types of imaging tests like X-rays, CT scans, and MRI scans can be used as biomarkers to measure the size of a tumor and to see if a drug may have helped the tumor shrink in size. CureGRIN is interested in investigating if there are ways that GRIN and other GRI disorders change the blood, cerebrospinal fluid, or other biological functions that can be reversed with treatments/cures. We want to find biomarkers that may be able to measure things such as NMDAR levels in megakaryocytes in the bone marrow and platelets in the blood, devices that measure and estimate the likelihood of a seizure occurring, and so many other biological processes related to GRIN and other GRI disorders.
The U.S. Food and Drug Administration (FDA) groups biomarkers into seven different categories including diagnostic, susceptibility/risk, prognostic, predictive, response, monitoring, and safety. Diagnostic biomarkers confirm the presence of a particular medical condition in order to diagnose patients. Susceptibility/risk biomarkers are used to assess someone’s risk of developing a medical condition. Prognostic biomarkers are measured at the baseline (before treatment) and are important for assessing the likelihood of progression of a medical condition or other future events. A predictive biomarker helps to find people who are more likely to respond to a particular treatment. A response biomarker is used to measure changes in physiological levels in the body as a result of receiving treatment and provides early information about if a treatment might be working. Monitoring biomarkers are used repeatedly to assess the progression of a medical condition or a response to treatment. A safety biomarker is used to make sure that a treatment does not cause adverse effects. The FDA also notes the importance of the analytical and clinical validation of biomarkers. Analytical validation confirms that a biomarker test or device is specific, accurate, and precise, whereas clinical validation confirms that a particular biomarker performs as it is supposed to. CureGRIN plans to explore different types of biomarkers that can help doctors and researchers understand more about GRIN and other GRI disorders and to help determine if treatments result in measurable clinical improvements.
Therefore, the question of finding and characterizing biomarkers will be important for CureGRIN to partner with doctors and researchers to assess how GRIN and other GRI disorders progress, how patients respond to treatments and cures as they are developed and tested, and perhaps to even diagnose patients earlier.
Reference: FDA-NIH Biomarker Working Group. BEST (Biomarkers, EndpointS, and other Tools) Resource [Internet]. Silver Spring (MD): Food and Drug Administration (US); 2016-. Available here. Co-published by National Institutes of Health (US), Bethesda (MD).
Essential Question 3: Is a cure possible at any age?
Each Thursday throughout the summer, CureGRIN will be sharing a blog post that summarizes one of the 10 “Essential Questions” that need to be answered in order to find cures and treatments for GRIN Disorder.
Today, we focus on Essential Question 3: Is a cure possible at any age?
We do not know exactly how or when interventions must be administered for GRIN and other GRI patients to have big improvements. Will a cure only be possible for young children or teens and adults too? In a study published in 2020, Dr. Amy Ramsey, a member of CureGRIN’s Scientific Advisory Board, and her team tested this question on mice with a GRIN1 mutation.
Specifically, Dr. Ramsey and her team developed a mouse model where adult GRIN1 loss-of-function mice could be “rescued” to wild-type using a gene editing technique with a type of on/off switch. This technique (called Cre recombinase) is unique to mice and gives researchers control over the timing and location of gene expression. This approach allowed the researchers to turn the mutation off when the mice reached adulthood and resulted in an increase in the level of NMDARs in the GRIN1-rescue mice. The GRIN1-rescue mice were able to gain strength, become more social, and perform cognitive assessments comparably to wild-type mice born with typical levels of GRIN1 gene expression. Therefore, this study suggests that developmental changes in NMDAR expression levels can be reversed and that this reversal can occur in adulthood. This means that even after the brain has completed development, there is the possibility for brain functioning to be restored.
Additional studies may be performed in the future on mice with variants in other genes and human patient variants to better understand if these other types of variants can also be restored in adulthood. Since this particular study was done in mice, more research also needs to be done to determine if the changes in the human brain of GRIN and other GRI patients can be cured at any age. Further research should be performed to address the questions of whether the deficits in the brain altered the circuitry permanently and has the human brain wired around deficits in such a way that reversal has unanticipated results? CureGRIN will be working with scientists and biotechnology companies around the world to explore the answers to these questions.
To learn more about the research study mentioned above, check out the article here , read our blog post and watch our video interview with Dr. Ramsey and Dr. Catharine Mielnik.
Essential Question 4: What’s the best delivery route for gene therapy?
Each Thursday throughout the summer, CureGRIN will be sharing a blog post that summarizes one of the 10 “Essential Questions” that need to be answered in order to find cures and treatments for GRIN Disorder.
Today, we focus on Essential Question 4: What’s the best delivery route for gene therapy?
Gene therapy is a technique that can be used to introduce genetic material to fix variants in genes or to make a protein. Researchers have a few ideas of the types of gene therapy medicines and strategies (e.g., Knockdown and Replace) that could possibly be created to treat GRIN and other GRI disorders, though it is important to note that no gene therapy medicines are currently available for GRIN or GRI disorders. Once a gene therapy medicine is developed for GRIN and other GRI disorders, understanding how we should deliver this medicine is extremely important. Should the medicine be delivered into the blood through a vein, into the spinal cord, or to a specific region of the brain?
The nervous system is very complex. Therefore, there are some challenges to delivering the ideal gene therapy. Some of these challenges include the blood-brain barrier (BBB) and the invasiveness of a particular delivery method.
The BBB acts as a wall between the brain’s blood vessels, or capillaries, and the other parts of the brain’s tissues. The skull and cerebrospinal fluid (CSF) keep the brain protected from physical damage, yet the BBB protects the brain from damage from toxins and pathogens in the blood. The BBB only lets in gases (such as oxygen), small molecules, and fat-soluble molecules. Bigger molecules, like glucose, can come in through a special way called transporter proteins.
The BBB is very good at keeping undesired things out of the brain, which poses some challenges for getting certain medicines into the brain. Advances in BBB delivery technology in the future will help to accelerate treatments, and there are also ways to deliver medicine that bypass the BBB.
Another challenge to delivering gene therapy is the invasiveness of a particular delivery method. There are a number of ways to deliver gene therapy. Some of these methods include intravenous, intrathecal, intracerebroventricular, and intraparenchymal administration.
Intravenous administration involves injecting the medicine into the vein. Intrathecal administration requires injecting the medicine into the spinal canal where the CSF is located. Intracerebroventricular administration requires injecting the medicine into the CSF in the brain. Intraparenchymal administration calls for injecting the medicine into gray matter tissue in the brain. As you can see, some of these delivery methods are more invasive than others. CureGRIN will be working with doctors, scientists, and biotechnology companies around the world to research these gene therapy delivery options to find the safest, least invasive option that results in getting the medicine to all the right places to help GRIN and GRI patients.
Essential Question 5: How can we deliver gene therapies for larger genes?
Each Thursday throughout the summer, CureGRIN will be sharing a blog post that summarizes one of the 10 “Essential Questions” that need to be answered in order to find cures and treatments for GRIN Disorder.
Today, we focus on Essential Question 5: How can we deliver gene therapies for larger genes?
There are many types of gene therapy medicines and strategies. Researchers have some ideas on certain gene therapy medicines and strategies that could possibly be developed to treat GRIN and other GRI disorders, though no gene therapy medicines are currently available for GRIN or other GRI disorders. The ideal gene therapy would entail only one medicine per gene, regardless of the specific variant. One type of gene therapy strategy that could be developed is called “Knockdown and Replace.”
“Knockdown and Replace” gene therapy would be delivered through the central nervous system or directly into the brain and would have two instructions. Firstly, it would tell the gene to stop making proteins from both copies of the patient’s genes. Secondly, it would introduce and boost a synthetic copy of the gene. The result would be that some mutant proteins could still possibly get made, but they would be vastly outnumbered by the wildtype proteins.
This strategy of gene therapy could be delivered using Adeno-Associated Virus Vectors (AAVs). There are currently two FDA approved AAV gene therapies. An AAV therapy called Luxturna was approved for a rare inherited retinal dystrophy in 2017, and an AAV therapy called Zolgensma was approved for spinal muscular atrophy in 2019.
AAVs are essentially carriers that are altered from a naturally occurring virus into a delivery tool for gene therapy. The viral genetic material is replaced with new genetic material. Then, the AAV is used to deliver wildtype copies of a gene to the right tissues or organs in the body.
AAVs have a carrying capacity of about 4.7kb of nucleotides. The “instructions” for “Knockdown and Replace” gene therapy contain approximately 1.1kb of code, meaning that the maximum size of a gene that can fit is approximately 3.6kb. The GRIN1 and GRIA genes likely fit into AAV. As seen in the table below, some of the GRIN genes are too big to fit into this type of vector.
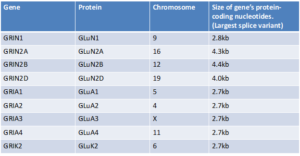
So, CureGRIN will be working with researchers to figure out how we can safely and effectively deliver gene therapies to patients with GRIN2A, GRIN2B, and GRIN2D Disorder. Some possible strategies to consider may include using dual AAVs or using other types of vectors such as adenovirus vectors and lentivirus vectors. We may also research additional gene therapy candidates like antisense oligonucleotides (ASOs), mRNA, and/or CRISPR. CureGRIN will partner with researchers and biotechnology companies to assess what the right strategy is for the delivery of gene therapy for GRIN2A, GRIN2B, and GRIN2D.
Essential Question 6: What are optimal drugs and molecules targeting NMDARs and related ion receptors?
Each Thursday throughout the summer, CureGRIN will be sharing a blog post that summarizes one of the 10 “Essential Questions” that need to be answered in order to find cures and treatments for GRIN Disorder.
Today, we focus on Essential Question 6:
What are optimal drugs and molecules targeting NMDARs and related ion receptors?
We’ve spent the last couple of weeks outlining the details of CureGRIN’s plans to research gene therapy strategies. However, this week, we are going to shift our focus to drugs and molecules.
The U.S. Food & Drug Administration (FDA) defines a drug as “a substance intended for use in the diagnosis, cure, mitigation, treatment, or prevention of disease.” However, the FDA classifies new molecules that haven’t yet been approved as a “new chemical entity” or a “new molecular entity.” Hence, when CureGRIN talks about drugs, we are describing medicines that have been previously approved by the FDA for another disease or condition but could still possibly be helpful for GRIN and other GRI disorder patients. However, when CureGRIN talks about molecules, we are describing potential medicines that haven’t yet been approved by the FDA that might help GRIN and other GRI disorder patients.
As a part of CureGRIN’s three-tiered approach for finding treatments and cures for GRIN and other GRI disorders, we plan to spend about 30% of our time and resources researching drugs and molecules that target NMDARs, AMPARs, and kainate receptors. These drugs and molecules include agonists, antagonists, PAMs, NAMs, etc. Can these various types of drugs bring GRIN-related receptors and other GRI-related receptors into balance?
First, let’s better understand what these types of drugs and molecules are, how they work, and how they might help.
Agonists are substances that bind to and activate a receptor, which produces a biological response. For example, glutamate is an agonist of the NMDAR. There are two types of agonists: endogenous agonists and exogenous agonists. Endogenous agonists are naturally produced in the body, such as hormones and neurotransmitters, whereas exogenous agonists are external factors, such as drugs.
An example of an NMDAR agonist is L-serine. L-serine has been studied in a patient with GRIN disorder with a GRIN2B loss-of-function variant. There were improvements in motor impairments, cognition, and communication in this patient. There is currently an ongoing clinical trial for L-serine, which is being led by Dr. Angels García Cazorla and the Barcelona GRIN team.
Antagonists are substances that interfere with or inhibit the action of the receptor. An example of an NMDAR antagonist is memantine. There have been several single-patient (N=1) studies where memantine was prescribed off-label.
These individual research studies have looked at patients with gain-of-function variants in the GRIN1 (Xu et al., 2021), GRIN2A (Pierson et al., 2014; Amador et al., 2020), and GRIN2B genes (Platzer et al., 2017). Some of these studies have shown that the memantine treatment reduced the frequency or burden of seizures in some of these individual patients. However, no large-scale clinical trials for memantine in GRIN disorder patients have been carried out.
PAMs (positive allosteric modulators) act to enhance the function of a receptor. PAMs work in the presence of an agonist (a substance, like glutamate, that activates a receptor), but they are not able to directly activate the receptor without the agonist. PAMs essentially increase an agonist’s ability to work, boosting the overall activity of the receptor.
Some of the classes of PAMs that have been investigated for NMDARs are spermine, pregnenolone sulfate, and 24 (S)-HC. 24 (S)-HC, also called SAGE 718, is now in clinical trials for Alzheimer’s and Parkinson’s Disease at Sage Therapeutics. The hurdle has been to find PAMs that target specific NMDAR subunit types (NR1, NR2A, NR2B, NR2D).
NAMs (negative allosteric modulators) are molecules that block the binding of the agonist (activator) to a receptor. A NAM binds to a site on the receptor that is different from the spot where the agonist binds, weakening the overall activity of the receptor. For example, Radiprodil is a drug that acts as a NAM of the NR2B subunit of the NMDA receptor. This drug has been investigated for a type of epileptic encephalopathy known as infantile spasm syndrome (ISS).
For each of these various types of drugs and molecules, it is important to note that it is crucial to have functional testing performed on a patient’s variant before deciding which drug options might work for them. We would expect agonists and PAMs to help patients with a clear loss-of-function variant, while antagonists and NAMs would help patients with a clear gain-of-function variant. If the wrong type of drug or molecule is given to a patient based on their functional testing, this could make their symptoms worse.
For example, if memantine (an NMDAR antagonist) was given to a patient with a loss-of-function variant, this would mean that their NMDARs would be working even less, which could result in increased seizure activity and other health complications. This is why memantine has only been prescribed for patients with gain-of-function GRIN gene variants.
In summary, there are many important factors to consider when thinking about drug development for GRIN and other GRI disorders. Therefore, CureGRIN will partner with researchers and pharmaceutical companies to assess what the right drugs and molecules are to help patients with GRIN and other GRI disorders.
Essential Question 7: Are there approved or late-stage drugs that could be repurposed for GRIN and related GRI Disorders?
Each Thursday throughout the summer, CureGRIN will be sharing a blog post that summarizes one of the 10 “Essential Questions” that need to be answered in order to find cures and treatments for GRIN Disorder.
Today, we focus on Essential Question 7:
Are there approved or late-stage drugs that could be repurposed for GRIN and related GRI Disorders?
Last week, we described our plans to research drugs and molecules that target NMDARs, AMPARs, and kainate receptors to help patients with GRIN and other GRI disorders. We discussed how different types of drugs and molecules including agonists, antagonists, PAMs, and NAMs might prove helpful for treating some patients. This week, we want to explore the idea that there could possibly be drugs out there already that might be able to help.
The U.S. Food & Drug Administration (FDA) has a very strict process for determining if drugs and molecules are safe and effective for the illness or disease that a drug is intended to treat, in order to minimize patient risk. This process requires researchers and pharmaceutical companies to conduct clinical trials and apply for approval through the Investigational New Drug (IND) application.
There are two ways that CureGRIN could possibly help in this area of the search for new treatments for GRIN and other GRI disorders.
Pharmaceutical companies have lots of drug candidates that did not previously meet the FDA’s standards and did not make it market. For example, if a medicine that was explored for a different disease but did not meet the criteria to be approved by the FDA, CureGRIN could perhaps encourage companies to explore this drug candidate for GRIN or other GRI disorders. However, we do not yet know of any such potential medicines.
CureGRIN could also partner with companies to do what’s called a high-throughput screening. A high-throughput screening is a process that can quickly scan through hundreds to thousands of molecules and compounds to help find drug targets. After a target is found, this leads to in vitro assays, where researchers in a laboratory study how the drug target works in cells, and in vivo assays, where researchers study how the drug target works in animal models (such as mice). If the drug target works well in cells and animal models, this could lead to clinical trials and potential FDA approval.
CureGRIN will partner with various researchers and pharmaceutical companies to assess all the drugs and molecules that could be available to help patients with GRIN and other GRI disorders.
Essential Question 8: Which symptoms are due to receptors outside of the brain?
Each Thursday throughout the summer, CureGRIN will be sharing a blog post that summarizes one of the 10 “Essential Questions” that need to be answered in order to find cures and treatments for GRIN Disorder.
Today, we focus on Essential Question 8: Which symptoms are due to receptors outside of the brain?
We know that the GRIN and GRIA genes are highly expressed in the brain. Yet, the GRIN genes are also expressed at low levels outside of the brain. The GRIA genes are expressed outside of the brain as well but to a much lower extent than GRIN genes. CureGRIN would like to explore the possibility that some of the symptoms of GRIN Disorder might be attributed to the expression of receptors outside of the brain. For example, is hypotonia caused by NMDARs in the brain or the somatic nervous system? Are respiratory issues caused by hypotonia originating in the brain neurons or localized in the respiratory system?
Gene expression involves the use of a molecule called messenger RNA (mRNA) to convert genetic information from genes into proteins. Scientists have been able to measure the amount of mRNA in different areas throughout the body. These studies have determined that the GRIN and GRIA genes have the highest expression levels in the brain.
One example of research on NMDARs outside of the brain is a study that was published in 2021, which assessed the expression of NMDARs in the human pulmonary artery (Dong et al.). The pulmonary artery works to carry deoxygenated blood from the heart to the lungs. Previous research has indicated that the NMDAR subunits GluN1 and GluN2A-D are expressed throughout the lungs and trachea. One cell type that expresses NMDARs in the lungs is the pulmonary airway smooth muscle cells.
When NMDARs are activated in these pulmonary airway smooth muscle cells, calcium (Ca2+) is released and the airway contracts. Yet, if there is too much activation of the NMDARs in the lungs, acute nitric oxide-dependent injury can occur. CureGRIN has been notified of a very small number of unexpected events related to respiratory issues in our patient community. Therefore, we are interested in learning more about how the NMDARs in these cells might play a role in GRIN disorder.
CureGRIN will be working with physicians and scientists around the world to explore what symptoms of GRIN Disorder might possibly be occurring because of receptors in other parts of the body.
Reference: Dong, Y.N., Hsu, FC., Koziol-White, C.J. et al. Functional NMDA receptors are expressed by human pulmonary artery smooth muscle cells. Sci Rep 11, 8205 (2021). https://doi.org/10.1038/s41598-021-87667-0 https://www.nature.com/articles/s41598-021-87667-0#citeas
Essential Question 9: Can we improve symptoms by targeting downstream?
Each Thursday throughout the summer, CureGRIN will be sharing a blog post that summarizes one of the 10 “Essential Questions” that need to be answered in order to find cures and treatments for GRIN Disorder.
Today, we focus on Essential Question 9:
Can we improve symptoms by targeting downstream?
Certain aspects of GRIN disorder and other GRI disorder symptoms could possibly be attributed to other factors in the body. In biology, downstream effects are things that affect or influence signaling later on in a pathway. CureGRIN wants to better understand which symptoms of GRIN and other GRI disorders can be ameliorated by combinations of substances that together target downstream effects including:
1) Oxidative stress
2) Neuroinflammation
3) Mitochondrial / nutrient signaling dysfunction (IGF, mTOR, etc.)
4) Excitatory / Inhibitory Imbalance
5) Methylation & other epigenetic markers
We will discuss each of these different types of downstream processes below.
Oxidative stress is a disproportion of free radicals (oxygen-containing molecules with electrons) and antioxidants in the body. Oxidative stress can lead to cell death and tissue damage.
Neuroinflammation is a basic immune response in the brain. A certain normal concentration of inflammation protects brain cells from damage, but excessive neuroinflammation intensifies damage.
Mitochondrial dysfunction occurs when the mitochondria (the so called “powerhouses of the cell”) don’t work as well as they should. This can be caused by exposure to certain environmental factors or genetic abnormalities.
Nutrient signaling involves various signaling pathways that are regulated by the availability of nutrients in the body. Different nutrient levels are required to activate signaling, which modulates cellular processes like metabolism. Insulin-like growth factor (IGF) is a hormone that promotes normal growth bone and tissue growth. Protein kinase mechanistic target of rapamycin (mTOR) is a cell growth regulator which is important for cellular metabolism and growth.
Epigenetic markers are like a light switch that tell our genes to turn on or off. There are different types of epigenetic markers such as chemical (methylation) or protein (histones). As a result of epigenetic markers, certain environmental factors like diet, prenatal nutrition, and stress can impact the genes that are passed on from one generation to the next.
We don’t yet know how all of these processes and downstream effects are affected in GRIN and GRI disorder patients. CureGRIN will be working with scientists around the world to explore what symptoms of GRIN and other GRI disorders might possibly be improved by targeting these downstream processes.
Essential Question 10: What are the functional and phenotypic details for each variant?
Each week throughout the summer, CureGRIN has shared a blog post that summarized one of the 10 “Essential Questions” that need to be answered in order to find cures and treatments for GRIN Disorder.
Today, we share the final blog post, where we focus on Essential Question 10:
What are the functional and phenotypic details for each variant?
Functional analysis is a type of test that is used to determine if a variant is Loss of Function (LoF) or Gain of Function (GoF). LoF variants are variants that result in a change to the gene product (protein) that has either less functioning than normal or no function. GoF variants are variants resulting in the gene product (protein) having either a stronger effect than normal or a different function. It is useful to know if a GRIN or other GRI gene variant is LoF or GoF because the mechanics of the variant is crucial to determine potential treatments. For improved diagnosis and effective treatment, we need a better understanding of the function of GRIN and other GRI gene variants.
CureGRIN wants to better understand the function of every GRIN or other GRI gene variant and what the resulting phenotypes (i.e., common traits and symptoms) are for each variant.
Numerous research groups are working to uncover the function of GRIN and other GRI gene variants. For example, Dr. Stephen Traynelis, a member of CureGRIN’s Scientific Advisory Board, leads the Center for Functional Evaluation of Rare Variants (CFERV) in Atlanta, GA, USA. One of CFERV’s many goals is to work to provide functional test results on all glutamate receptor variants (GRIN, GRIA, GRIK, and GRID) to stratify patients and ultimately understand subgroups for natural history (i.e., the expected course of a disorder). In 2020, CureGRIN awarded a $75,000 grant to CFERV to cover the cost of functional analysis for 50 patients with GRIN Disorder.
Additionally, the Barcelona GRIN team, which includes Dr. Xavier Altafaj, a member of CureGRIN’s Scientific Advisory Board, runs the GRIN variants database. The GRIN variants database aims to provide functional annotation of GRIN variants and compile all existing annotations of GRIN variants.
Furthermore, Dr. Johannes Lemke, a member of CureGRIN’s Scientific Advisory Board, leads the GRIN database. The GRIN database aims to provide data on variants, functions, and phenotypes for GRIN1, GRIN2A, GRIN2B patients.
Dr. Lemke, Dr. Traynelis, and colleagues are also involved in a new project called the GRIN Portal, which aims to provide information on GRIN Disorder and describe variant interpretation and classification. Other goals of the GRIN Portal include recruiting patients for the worldwide GRIN registry, visualizing registry data, and connecting researchers, clinicians, and families.
The GRIN Registry is essential for capturing patient phenotype information. Dr. Lemke leads the Europe, Asia, Africa and other chapter of the GRIN Registry, whereas Dr. Tim Benke, another member of CureGRIN’s scientific advisory board, as well as Dr. Kristen Park and Dr. Jennifer Bain, lead the North America, South America, and Australia chapter. Both groups are collecting data on genetic variants, clinical history, family history, and epilepsy features and are merging the data.
The data collected from the GRIN Registry will be important for conducting natural history studies. Natural history studies involve collecting information about the course of a particular disease. Moreover, natural history studies follow the disease progression from the onset of the disease until the disease is cured or treated. The FDA notes that natural history studies are helpful for developing safe and efficacious treatments for rare diseases, such as GRIN and other GRI disorders.
CureGRIN plans to continue to support researchers conducting functional analysis studies and phenotypic studies on identified variants across the GRI genes. Additionally, we do not know exactly how the brain’s architecture is changed by GRIN and other GRI gene variants. CureGRIN plans to partner with researchers and clinicians to do further analysis on existing animal models, perform natural history studies, and establish a brain donation program for GRIN Angels, to better understand changes in the human brain.
